Unit 6 Part I: The Mole
|
1/24-25/24
TEKS: C.8.A, C.8B and C.8C Objectives: The student will be able to: a. Define and use the concept of mole b. Calculate the number of atoms or molecules in a sample of material using Avogadro’s number. 1. EXCITING DEMOS Sodium and Water Combustion of Isopropyl Alcohol Ammonium Dichromate Decomposition Lead III Nitrate With Potassium Iodide 2. Dimensional Analysis Process 3. What is the mole and what are representative particles? 4. RP to Mole and Mole to RP Conversion Practice Homework: Complete the lab questions on SCHOOLOGY |
|
|
1/30-31/24
TEKS: C.8.A, C.8B and C.8C Objectives: The student will be able to: a. Define and use the concept of mole b. Calculate the number of atoms or molecules in a sample of material using Avogadro’s number. c. Calculate percent composition of compounds. 1. Spot Check RP Conversion work-QUESTIONS? 3. LESSON: Mole to Mass and Mass to Mole One Step Conversions TRY-IT: ONE STEP EQUATIONS AND MULTI-STEP EQUATIONS Homework: Complete gummy bear lab questions |
2/1-2/24
TEKS: C.8.A, C.8B and C.8C
Objectives: The student will be able to:
a. Define and use the concept of mole
b. Calculate the number of atoms or molecules in a sample of material using Avogadro’s number.
c. Calculate percent composition of compounds.
1. Spot check conversions assignment and submit
2. Decomposition of KClO3 with Gummy Bear
3. Copper II Chloride LAB (PART I ONLY)
Homework: Work on LAB QUESTIONS FOR Decomposition of KClO3 with Gummy Bear
TEKS: C.8.A, C.8B and C.8C
Objectives: The student will be able to:
a. Define and use the concept of mole
b. Calculate the number of atoms or molecules in a sample of material using Avogadro’s number.
c. Calculate percent composition of compounds.
1. Spot check conversions assignment and submit
2. Decomposition of KClO3 with Gummy Bear
3. Copper II Chloride LAB (PART I ONLY)
Homework: Work on LAB QUESTIONS FOR Decomposition of KClO3 with Gummy Bear
2/5-6/24
TEKS: C.8.A, C.8B and C.8C
Objectives: The student will be able to:
a. Define and use the concept of mole
b. Calculate the number of atoms or molecules in a sample of material using Avogadro’s number.
c. Calculate percent composition of compounds.
1. Copper II Chloride LAB PART II
2. Gallery WALK (TEAM ACTIVITY)
Homework: Continue with Copper II Chloride LAB Questions
TEKS: C.8.A, C.8B and C.8C
Objectives: The student will be able to:
a. Define and use the concept of mole
b. Calculate the number of atoms or molecules in a sample of material using Avogadro’s number.
c. Calculate percent composition of compounds.
1. Copper II Chloride LAB PART II
2. Gallery WALK (TEAM ACTIVITY)
Homework: Continue with Copper II Chloride LAB Questions
2/7/24 PT CONFERENCES 1/2 DAY: Continue with LAB Questions
2/8/24 TSI TESTING: Continue with LAB Questions
2/8/24 TSI TESTING: Continue with LAB Questions
2/9-12/24 B DAy/A-DAY
1. GO OVER ANY QUESTIONS ON LABS: TURN IN!
2. Unit 6 QUIZ
Homework: NONE-unless you did not turn in lab
1. GO OVER ANY QUESTIONS ON LABS: TURN IN!
2. Unit 6 QUIZ
Homework: NONE-unless you did not turn in lab
|
2/13/24 B DAY
TEKS: C.8.A, C.8B and C.8C Objectives: The student will be able to: a. Define and use the concept of mole b. Calculate the number of atoms or molecules in a sample of material using Avogadro’s number. c. Calculate percent composition of compounds. 1. Hydrate NOTES with practice problems 2. Hydrate LAB (ONE LAB FOR ALL TEAM MEMBERS) Homework: Individual Hydrate Practice Problems |
Feb. 14, 2024 A-DAY HALF-DAY I WILL ONLY SEE 2nd Period (WE WILL NOT CONTINUE CONTENT-MENTAL HEALTH DAY)
Feb. 15, 2024 B-DAY TSI DAY: I will only see 7th (WE WILL NOT CONTINUE CONTENT-MENTAL HEALTH DAY)
Feb. 15, 2024 B-DAY TSI DAY: I will only see 7th (WE WILL NOT CONTINUE CONTENT-MENTAL HEALTH DAY)
Feb. 16-20, 2024
TEKS: C.8.A, C.8B and C.8C
Objectives: The student will be able to:
a. Define and use the concept of mole
b. Calculate the number of atoms or molecules in a sample of material using Avogadro’s number.
c. Calculate percent composition of compounds.
1. Hydrate NOTES with practice problems
2. Hydrate LAB (ONE LAB FOR ALL TEAM MEMBERS): THIS WILL TAKE MULTIPLE DAYS
Homework: Hydrate Practice Problems (4)
TEKS: C.8.A, C.8B and C.8C
Objectives: The student will be able to:
a. Define and use the concept of mole
b. Calculate the number of atoms or molecules in a sample of material using Avogadro’s number.
c. Calculate percent composition of compounds.
1. Hydrate NOTES with practice problems
2. Hydrate LAB (ONE LAB FOR ALL TEAM MEMBERS): THIS WILL TAKE MULTIPLE DAYS
Homework: Hydrate Practice Problems (4)
Feb. 21-22, 2024
TEKS: C.8D
Objectives:
The students will be able to:
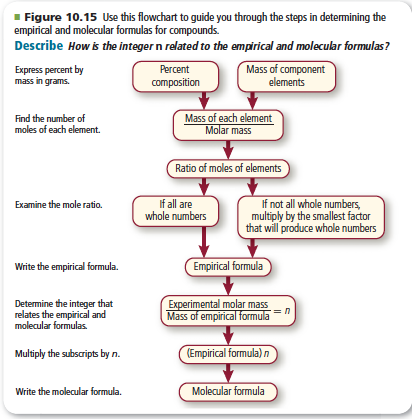
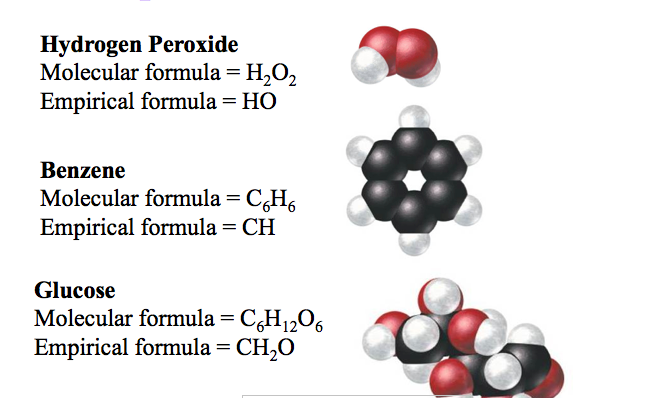
Differentiate between empirical and molecular formulas and derive the empirical and molecular formula
1. Empirical and Molecular Formula LESSON with practice
Homework: REVIEW NOTES
SMALL QUIZ COMING UP SOON ON EMPIRICAL AND MOLECULAR FORMULAS AND HYDRATES
TEKS: C.8D
Objectives:
The students will be able to:
Differentiate between empirical and molecular formulas and derive the empirical and molecular formula
1. Empirical and Molecular Formula LESSON with practice
Homework: REVIEW NOTES
SMALL QUIZ COMING UP SOON ON EMPIRICAL AND MOLECULAR FORMULAS AND HYDRATES
2/23-26/24
Refresh HYDRATES and E/M
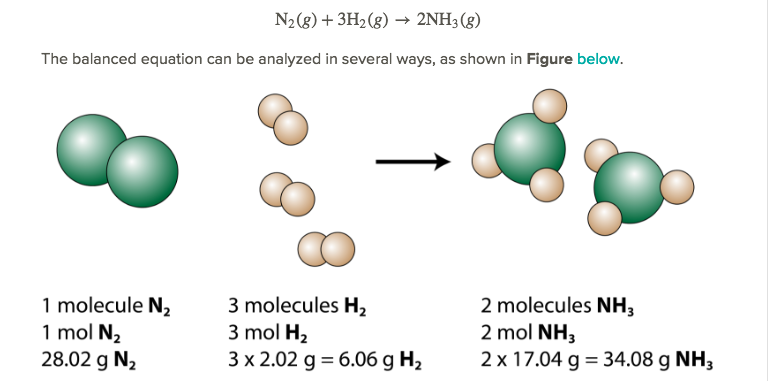
STOICHIOMETRY BIG LESSON NOTES
Homework: REVIEW NOTES
Refresh HYDRATES and E/M
STOICHIOMETRY BIG LESSON NOTES
Homework: REVIEW NOTES
2/27-2/28/24
TEKS: C.8.E, C.8G, C.8H
Objectives: The student will be able to:
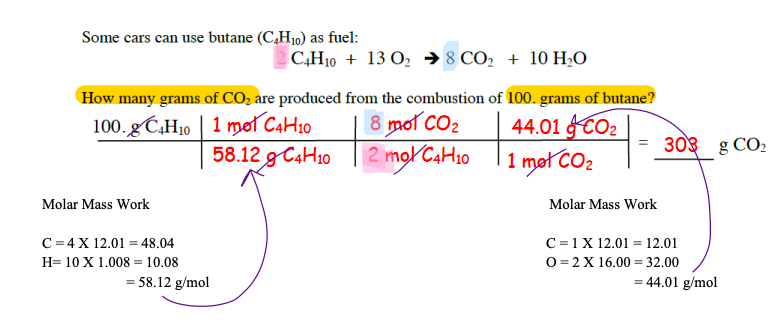
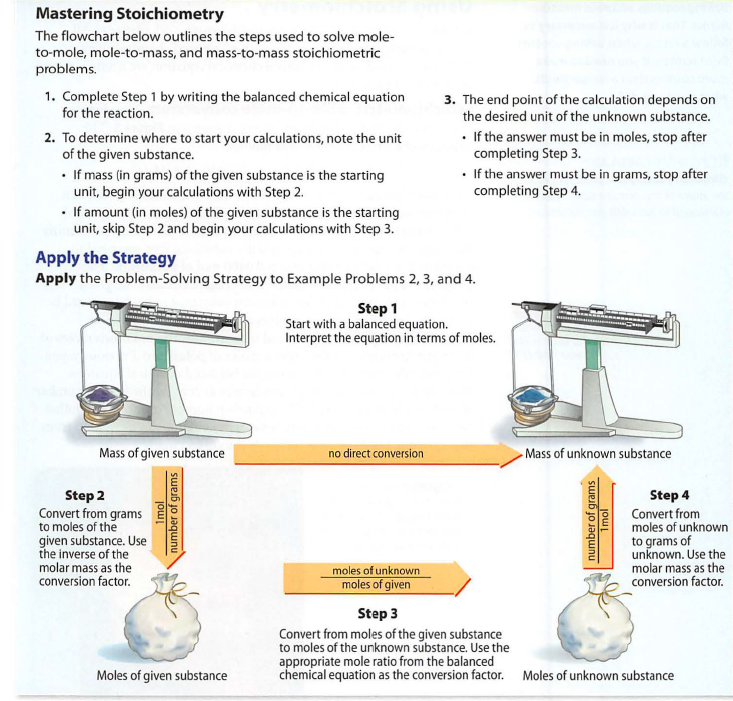
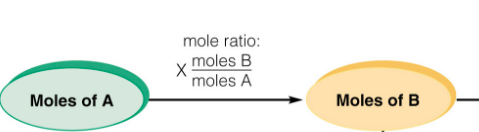
Perform three levels of stoichiometric operations: Mole-Mole, Mole-Mass, Mass-Mass
TEKS: C.8.E, C.8G, C.8H
Objectives: The student will be able to:
Perform three levels of stoichiometric operations: Mole-Mole, Mole-Mass, Mass-Mass
1. Need Independent Practice?! GO FOR IT!
2. Stoichiometry by Color! FOLLOW THE NOTES! Divide and Trust! Partner up with ONE other person. Each person completes 5.
NOTE: Coloring the actual fish is optional.
NOTE: Coloring the actual fish is optional.
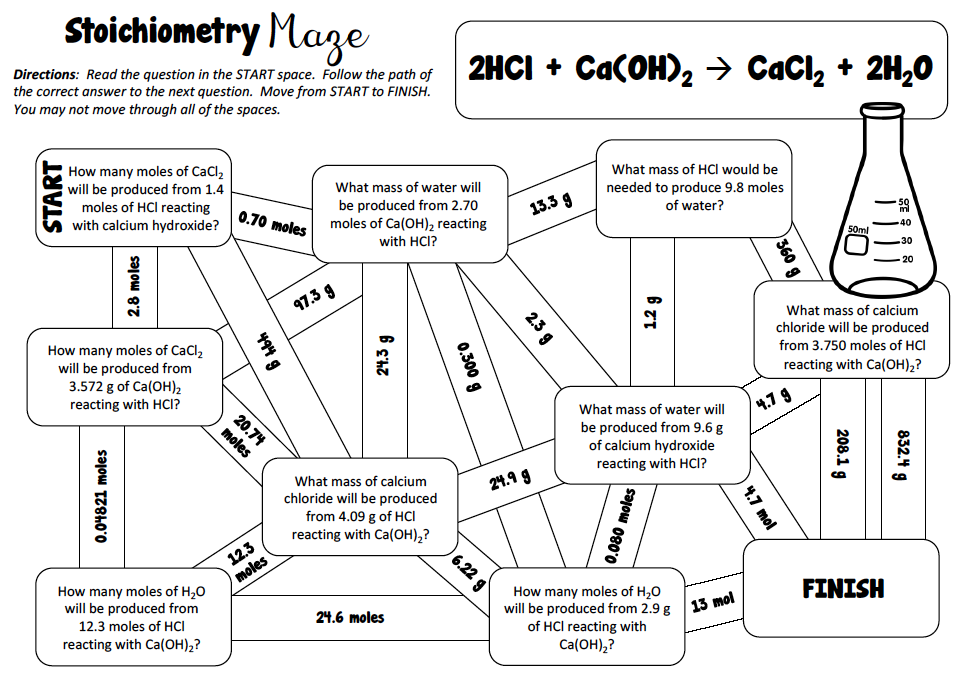
3. Stoichiometry MAZE
2/29-3/1/24
UNIT 6 PART II QUIZ and UNIT 7 STOICHIOMETRY (NINE WEEKS ENDS)
UNIT 6 PART II QUIZ and UNIT 7 STOICHIOMETRY (NINE WEEKS ENDS)
SPRING BREAK INTERCESSION
Unit 7 : Stoichiometry REFRESHER
3/18-19/24
1. Complete the warm-up question you feel comfortable with. THIS IS THE FINAL REVIEW FOR THE THREE LEVELS OF STOICHIOMETRY...
1. Complete the warm-up question you feel comfortable with. THIS IS THE FINAL REVIEW FOR THE THREE LEVELS OF STOICHIOMETRY...
Click Black Buttons open the document!
2. Gram-Gram Stoichiometry Murder FOUR suspects (4 problems): each table partner solves 1/4 problems and then the team determines the person who committed the murder.
2. Gram-Gram Stoichiometry Murder FOUR suspects (4 problems): each table partner solves 1/4 problems and then the team determines the person who committed the murder.
2. Limiting Reactant, Percent Yield and Theoretical Yield, NEW NOTES (while we wait for lab product to form)
3/20-21/24
1. (Copper and Silver Nitrate) Pre-lab and Procedure
2. While we wait 30 minutes....Limiting Reactant, Percent Yield and Theoretical Yield Assignment QUESTIONS?
HOMEWORK: COMPLETE POST LAB CALCULATIONS: YOUR DRY RESULTS WILL BE POSTED FOR YOU TO USE SO YOU CAN FINISH LAB
1. (Copper and Silver Nitrate) Pre-lab and Procedure
2. While we wait 30 minutes....Limiting Reactant, Percent Yield and Theoretical Yield Assignment QUESTIONS?
HOMEWORK: COMPLETE POST LAB CALCULATIONS: YOUR DRY RESULTS WILL BE POSTED FOR YOU TO USE SO YOU CAN FINISH LAB
3/22-25/24
1. Wrap up LAB data and questions
2. Stoichiometry: How is cider making like stoichiometry?
1. Wrap up LAB data and questions
2. Stoichiometry: How is cider making like stoichiometry?
3/26-27/24
1. Does changing the amount of reactant 1 used (0.5 g, 1.0g, 2.5g, 5.0g) affect the amount of product formed if reactant 2 remains constant? Will there at one point be too much of one reactant and not enough of the other?
Homework: Complete LAB Write-UP
1. Does changing the amount of reactant 1 used (0.5 g, 1.0g, 2.5g, 5.0g) affect the amount of product formed if reactant 2 remains constant? Will there at one point be too much of one reactant and not enough of the other?
Homework: Complete LAB Write-UP
1. (Watch Video, Read Caption Below and Read article) Write a 400-500 page essay on the topic below.
https://www.youtube.com/watch?v=Y2sjYOGSV7E
Explain in detail how airbags work using both chemistry and general physics principles.
Explain how an airbag is an example of an application for stoichiometry.
SMALL PROJECT: Create a Digital Graphic Organizer or Digital Brochure
Brochure/GO must meet all requirements posted as well as be neat/organized, have flow, contain images that support content, and be creative and colorful. Words cannot be copied directly. Write in your own words without losing the scientific facts.
A: Properties (can look up online)
* Physical Properties and Chemical Properties
* Value of silver per gram
B. History and extraction and processing of silver (Britannica Button ONLY)
* Basic History
* Main mining locations
*Detailed Extraction and Refining Processes (Britannica Button ONLY)
C: Application w/Explanation 0f Silver's use in THREE industrial fields (click link)
https://www.silverinstitute.org/silver-in-industry/
D: THREE "Did you know that...? " statements based on the Silver News Articles (Silver News (Button ONLY):
F. Talking Silver Podcast: Summary (click link)
https://podcasts.apple.com/us/podcast/talking-silver/id1568101851
E: Citations APA Format
END OF UNIT 7